|
Amyotrophic Lateral Sclerosis (ALS) is a neurological disease which impacts motor neurons that are involved in muscle movement throughout the body.īrainStorm Cell Therapeutics an Israeli biotechnology company developing autologous stem cell therapies for neurodegenerative diseases has developed adult stem cell treatment for patients with ALS. 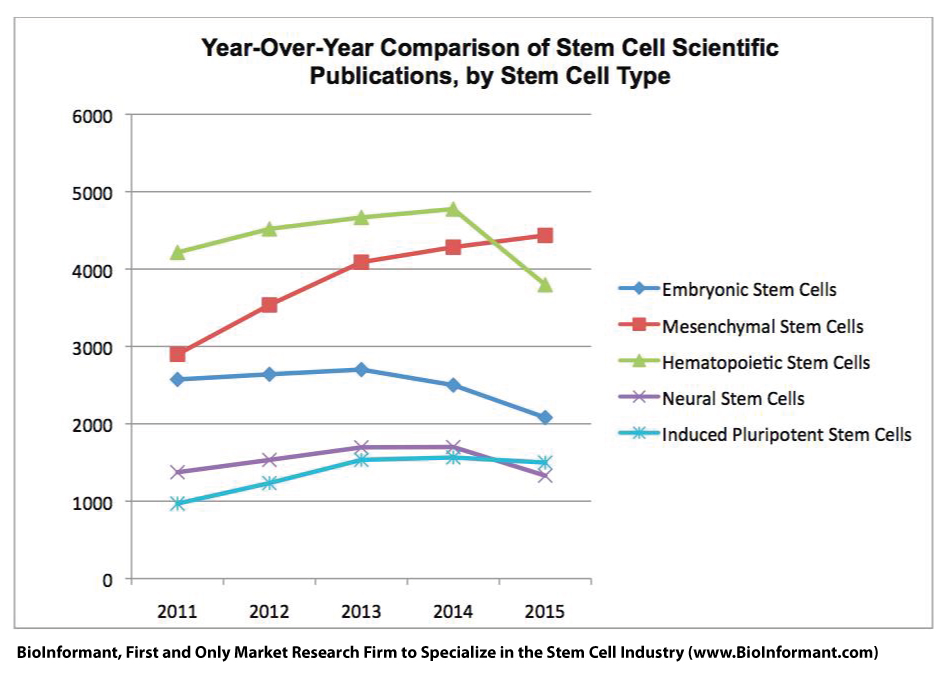
The disease is no respecter of persons and British physicist Stephen Hawking is perhaps its most famous living victim. Muscle cramps and twitches are also closely associated with ALS. These symptoms are wide-ranging and include progressive muscle weakness, unexplained fatigue of the arms and legs, slurred speech, tripping and dropping things. Treatment for ALS has so far recorded modest successes in slowing the progress of the disease and in managing the symptoms. Stem Cell Therapy for Amyotrophic Lateral Sclerosis may soon be a reality in many countries of the world. ALS is invariably fatal and the race to find a cure has been ongoing since the disease was first identified by French neurologist Jean-Martin Charcot in 1869. Brainstorm's share price dropped 66% Tuesday morning, to trade at about $4.Amyotrophic Lateral Sclerosis (ALS), also known as Lou Gehrig’s disease is a rapidly progressive neurological disease that causes dysfunction of the nerves that control muscle movement. that this clearly has a pathway - probably more than one pathway - for an approval without doing another trial," Lebovits said. "We do feel, all of us in Brainstorm reviewing the data. On average, early ALS patients treated with the NurOwn cell therapy declined 1.77 points on the functional scale over the trial's 28-week testing period, which was better than the 3.78 point decline seen among their placebo-treated counterparts. "In other words, we saw more variability in the measurement of ALS symptoms and function at the lower end of the functional scale," he said.ĭespite the setback, Brainstorm executives say there's enough positive data behind the NurOwn therapy to keep moving it forward in ALS.įor example, a pre-specified analysis of participants with early ALS found a much greater difference in response rates, 35% versus 16%, between those who got NurOwn and placebo. 
Ralph Kern, Brainstorm's chief medical officer, said one possible reason for the trial's high placebo response could be the number of participants with severe ALS, which was larger than in other ALS studies and more than what Brainstorm had expected. Kern said these patients are often less responsive to treatment and harder to measure responses for - factors that may have weighed on the results. "We didn't hit the real home run we wanted," Lebovits said on a Tuesday morning call with investors. However, the response rate in the placebo group clocked in much higher than Brainstorm anticipated - at 28% - which ultimately led to the trial not succeeding. Indeed, the results presented Tuesday show a 35% response rate in NurOwn-treated patients. Based on those results, as well as historical data from other ALS trials, the company designed a late-stage trial that assumed 35% of the NurOwn-treated patients and 15% of placebo-treated patients would hit its primary goal: a 1.25-point improvement each month on a widely-used scale for measuring the function in ALS patients. In 2016, Brainstorm's cell therapy scored positive results in a medium-sized study of almost 50 ALS patients. 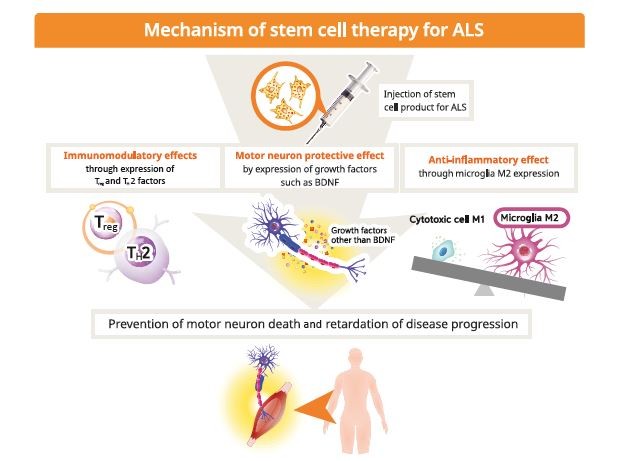
NurOwn takes stem cells from a patient's bone marrow, engineers them to secrete molecules that help protect and grow neurons, and then injects them back into the patient in the hope that they can slow or stop neurodegeneration. Currently, treatment is limited to just two medicines, both of which have their limitations.īrainstorm, founded in 2000, has been trying to add to that list with its so-called NurOwn technology platform. Like most neurodegenerative diseases, ALS has stumped the world's most skilled researchers and powerful drug companies.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
